 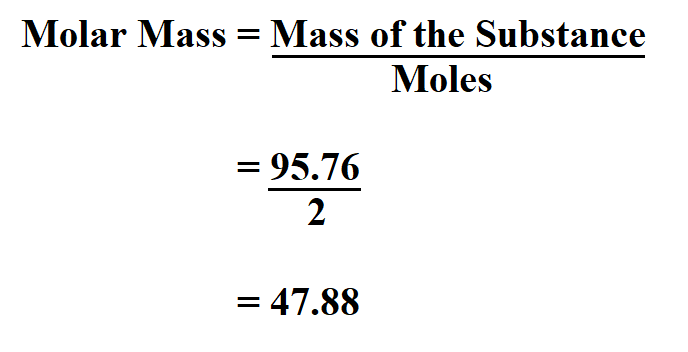 
In an earlier chapter, we described the development of the atomic mass unit, the concept of average atomic masses, and the use of chemical formulas to represent the elemental makeup of substances. Understanding the relationship between the masses of atoms and the chemical formulas of compounds allows us to quantitatively describe the composition of substances. For example, Dalton’s atomic theory was an attempt to explain the results of measurements that allowed him to calculate the relative masses of elements combined in various compounds. We can argue that modern chemical science began when scientists started exploring the quantitative as well as the qualitative aspects of chemistry. Define the amount unit mole and the related quantity Avogadro’s numberĮxplain the relation between mass, moles, and numbers of atoms or molecules, and perform calculations deriving these quantities from one another.Calculate formula masses for covalent and ionic compounds.Since we are converting grams into moles, we need to flip this conversion factor so that grams are on the bottom in order to cancel.ħ13 g x 1 mol Al 2(SO 4) 3/342.17 g = 2.Learning Objectives By the end of this section, you will be able to: The molar mass of Al 2(SO 4) 3 was earlier calculated to be 342.17 g/mol. How many moles are in 713 grams of Al 2(SO 4) 3? We earlier calculated the molar mass of Fe 2O 3 to be 159.70 g/mol.ģ.81 mol Fe 2O 3 x 159.70 g/1 mol Fe 2O 3 = 608 gĤ. What is the mass in grams of 3.81 moles of Fe 2O 3? The following two example problems will illustrate how to use molar mass as a conversion factor between moles and mass. This allows us to convert between grams of Al 2(SO 4) 3 and moles of Al 2(SO 4) 3. Just like there are 12 inches/1 foot, for Al 2(SO 4) 3 there are 342.17 g/mol. This looks a lot like a conversion factor that relates two units: grams to moles. Notice that the units on molar mass are grams per mole (g/mol). There are 5 Mn atoms (mass of 54.94) and 16 O atoms (mass of 16.00). Or Avogadro’s number, 6.022×10 23, of Fe 2O 3 formula units (ionic compound particles) would have a mass of 159.70 g. So one mole of Fe 2O 3 would have a mass of 159.70 g. To calculate its molar mass we have to add up the mass of two iron atoms and three oxygen atoms. To find the mass of one mole of any substance we count the elements present and add up their atomic masses.įor example, let’s calculate the molar mass of iron(III) oxide, Fe 2O 3. Fe 2O 3 is composed of two iron atoms and three oxygen atoms. So one mole of hydrogen, or 6.022×10 23 hydrogen atoms, has a mass of 1.01 g. Remember that one mole is equal to Avogardo’s number, 6.022×10 23 particles. But one incredibly handy aspect of the periodic table is that those same masses are also the mass of one mole of the element in grams.įor example, one atom of hydrogen has a mass of 1.01 amu, but one mole of hydrogen has a mass of 1.01 g. For example, the atomic mass of hydrogen (symbol H) is 1.01 amu (atomic mass units). We saw in a previous lesson on atomic mass that the periodic table provides us with the average atomic mass of elements. Molar mass is literally the mass of one mole of a substance. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
